PHILADELPHIA -- A new treatment for patients with a form of congenital retinal blindness has shown success in improving vision, according to results published today in Nature Medicine led by researchers at the Scheie Eye Institute in the Perelman School of Medicine at the University of Pennsylvania.
The treatment was designed for patients with CEP290 mutations and a diagnosis of Leber congenital amaurosis (LCA). CEP290 acts as a gate between two compartments of photoreceptor cells – a type of sensory neuron in the retina that converts light into signals that provide vision. Primary photoreceptor blindnesses - such as those caused by CEP290 mutations - are the most common form of LCA and they are currently not treatable.
In this clinical study at sites in the United States and Europe, participants received an intraocular injection of an oligonucleotide – a short RNA molecule – created to reduce the mutant CEP290 protein levels in the photoreceptors and restore retinal function.
“It was very dramatic to see one of the patients improve from only being able to differentiate light or dark to reading many letters on an eye chart at two months following the first injection,” said lead author Artur V. Cideciyan, PhD, a research professor of Ophthalmology. “So, we performed a thorough interim analysis of all results from all patients.”
Ten patients received at least one injection into their worst-seeing eye. At three months after the first injection, half of the patients showed improvements in visual acuity, measured by the ability to either read letters, or distinguish direction of black and white bars.
The most basic functions of photoreceptors are to receive and then signal light. This aspect of vision was tested in patients by presenting flashes of light in the dark and measuring the dimmest flash intensity detected. Treated eyes could detect on average more than six-fold dimmer lights three months after the injections compared to before. The results were highly statistically significant. Evaluations with two colors of flashes suggested that it was the cone photoreceptors used for daytime vision that were improving with the treatment.
“This therapy to repair the genetic defect in LCA10 is a breakthrough in treatment strategy and will open the door for clinical trials in other patients with this condition and other similar conditions that are currently untreatable,” said Samuel G. Jacobson, MD, PhD, a professor of ophthalmology and co-lead author of the study.
All patients enrolled so far into the study had two different CEP290 mutations. The p.Cys998X mutation causing a splicing defect was common in all patients and was specifically targeted by the oligonucleotide.
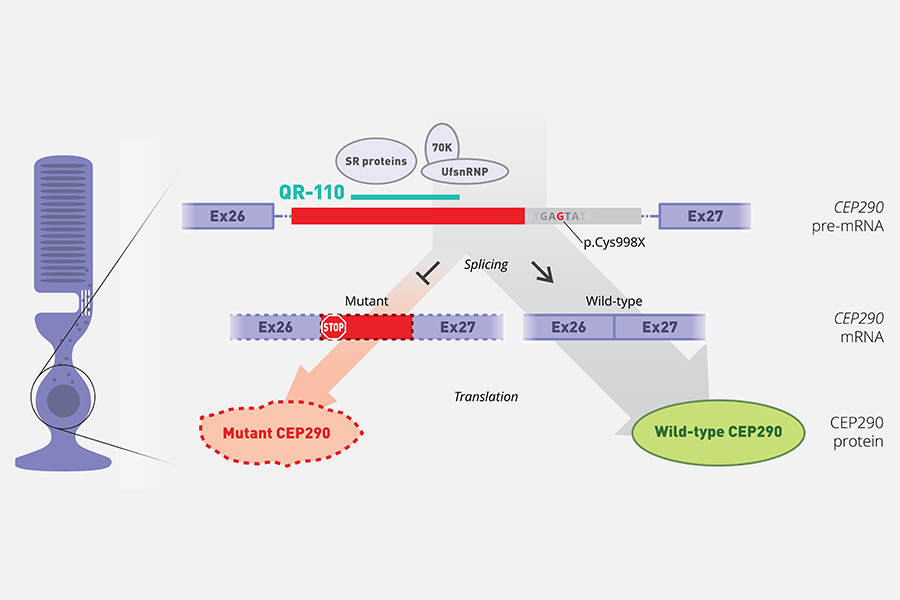 QR-110 Mechanism of Action: In wild-type cells, splicing results in correct transcripts and therefore normal functional protein. In cells carrying the p.Cys998X mutation a cryptic splice site is also recognized. Thus two types of transcripts are produced: a mutant transcript, and a wild-type transcript. When QR-110 is present, the recognition of the cryptic splice site is blocked, and the production of wild-type transcripts is favored.
QR-110 Mechanism of Action: In wild-type cells, splicing results in correct transcripts and therefore normal functional protein. In cells carrying the p.Cys998X mutation a cryptic splice site is also recognized. Thus two types of transcripts are produced: a mutant transcript, and a wild-type transcript. When QR-110 is present, the recognition of the cryptic splice site is blocked, and the production of wild-type transcripts is favored.
“We performed an extreme form of personalized medicine where we targeted not just a specific gene but a specific mutation in a gene,” said Cideciyan. “It is gratifying to see clinical evidence supporting the action of this oligo predicted from basic science. In the future, we hope to evaluate patients carrying two identical splicing mutations to determine whether the efficacy of the injection might be even greater.”
The authors note that the clinical trial is ongoing to measure safety and efficacy of the injections long-term and the effects of further doses.
Additional Penn authors include Jason Charng, Alexandra V. Garafalo, Alejandro J. Roman, and Alexander Sumaroka. Other authors include Arlene V. Drack, Ian C. Han, Maria D. Hochstedler, Wanda L. Pfeifer, Elliott H. Sohn and Stephen R. Russell from Carver College of Medicine, University of Iowa; Allen C. Ho from Wills Eye Hospital, Thomas Jefferson University; Magali Taiel, Michael R. Schwartz, Patricia Biasutto, Wilma de Wit, Peter Adamson, David M. Rodman, Gerard Platenburg, and Maria D. Tome from ProQR Therapeutics; Michael E. Cheetham from UCL Institute of Ophthalmology, and Irina Balikova, Fanny Nerinckx, Julie De Zaeytijd, Caroline Van Cauwenbergh, and Bart P. Leroy from Ghent University.
The trial is being funded by ProQR Therapeutics.
Penn Medicine is one of the world’s leading academic medical centers, dedicated to the related missions of medical education, biomedical research, excellence in patient care, and community service. The organization consists of the University of Pennsylvania Health System and Penn’s Raymond and Ruth Perelman School of Medicine, founded in 1765 as the nation’s first medical school.
The Perelman School of Medicine is consistently among the nation's top recipients of funding from the National Institutes of Health, with $550 million awarded in the 2022 fiscal year. Home to a proud history of “firsts” in medicine, Penn Medicine teams have pioneered discoveries and innovations that have shaped modern medicine, including recent breakthroughs such as CAR T cell therapy for cancer and the mRNA technology used in COVID-19 vaccines.
The University of Pennsylvania Health System’s patient care facilities stretch from the Susquehanna River in Pennsylvania to the New Jersey shore. These include the Hospital of the University of Pennsylvania, Penn Presbyterian Medical Center, Chester County Hospital, Lancaster General Health, Penn Medicine Princeton Health, and Pennsylvania Hospital—the nation’s first hospital, founded in 1751. Additional facilities and enterprises include Good Shepherd Penn Partners, Penn Medicine at Home, Lancaster Behavioral Health Hospital, and Princeton House Behavioral Health, among others.
Penn Medicine is an $11.1 billion enterprise powered by more than 49,000 talented faculty and staff.