| > |
Researchers at the Abramson
Family Cancer Research Institute of the University of Pennsylvania have found that deleting a gene important in embryo development
leads to premature aging and loss of stem cell reservoirs
in adult mice. |
| > |
This gene, ATR, is essential for the
body’s response to damaged DNA, and mutations in proteins
in the DNA damage response underlie certain types of cancer
and other disorders in humans. |
| > |
In the current study, the mouse cells
without ATR had an overwhelming amount of DNA damage
and could not contribute to tissue renewal; nevertheless,
the 10 to 20 percent of cells that escaped ATR deletion
were able to reconstitute tissues in the engineered mice,
at least initially. However, it appears that in the long
run, even these cells were insufficient to maintain tissue
integrity. |
| > |
This work appears in the inaugural issue
of Cell Stem Cell. |
(PHILADELPHIA) – Researchers at the Abramson
Family Cancer Research Institute of the University of Pennsylvania have found
that deleting a gene important in embryo development leads to premature
aging and loss of stem
cell reservoirs in adult mice. This gene,
ATR, is essential for the body’s response to damaged DNA,
and mutations in proteins in the DNA damage response underlie certain
types of cancer and other disorders in humans. This work appears
in the inaugural issue of Cell Stem Cell.
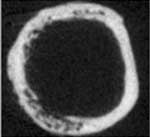 |
ATR deletion leads to cortical (the dense
surface part of bone) bone loss.
Click on thumbnail
to view full-size images |
“The reason these mice age prematurely is that we’re
exhausting their ability to renew tissues,” says Eric
J. Brown, PhD, Assistant Professor of Cancer Biology. “These
findings may be helpful to the aging and oncology fields since
premature aging syndromes and many cancers involve the loss of
DNA repair genes.”
When the researchers deleted ATR in the tissues of adult mice,
they noticed that the mice showed signs of premature aging, such
as hair
graying, hair
loss, and osteoporosis, within three to four
months.
To be able to renew itself, most tissues have a reservoir of specific
adult stem cells. These stem cells don’t divide as frequently
as other cell types since they need to maintain the integrity of
their DNA, and multiple divisions lead to natural breaks in DNA.
But when these stem cells are needed, their progeny can rapidly
divide and are able to replenish the tissue with new cells.
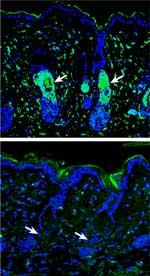 |
Hair follicle stem cells (green, top) are
lost (below) from follicles during hair regeneration
leading to alopecia, or hair loss.
Click on thumbnail
to view full-size images |
Brown explains that in the current study the mouse cells without
ATR had an overwhelming amount of DNA damage and could not contribute
to tissue renewal; nevertheless, the 10 to 20 percent of cells
that escaped ATR deletion were able to reconstitute tissues in
the engineered mice, at least initially. However, it appears that
in the long run, even these cells were insufficient to maintain
tissue integrity.
“Think of aging as a slow loss of stem cells, a deterioration
of pools of cells that reside in each tissue type,” says
Brown. “We accelerated the aging process by wiping out a
large fraction of these cells prematurely, in one fell swoop. Essentially,
ATR-deleted mice start their young adulthood with two strikes against
them in terms of long-term tissue maintenance, and so, they subsequently
age before their time.”
With this new knowledge of how DNA repair, stem cells, and the
aging process interconnect, Brown, first author Yaroslava
Ruzankina,
and Amma
Asare are currently using this mouse model to discover
compounds that preserve stem cells and may, consequentially, suppress
aging.
This work was funded in part by the National
Institute on Aging,
the Abramson Family Cancer Research Institute, and the General
Motors Cancer Research Scholars Program. Co-authors in addition
to Brown are Ruzankina, Asare, Carolina Pinzon-Guzman, Tony Ong,
Laura Pontano, George Cotsarelis, Valerie Zediak, Marielena Velez,
and Avinash Bhandoola, all from Penn.
###
PENN Medicine is a $2.9 billion enterprise
dedicated to the related missions of medical education, biomedical
research, and high-quality patient care. PENN Medicine consists
of the University of Pennsylvania School of Medicine (founded in
1765 as the nation's first medical school) and the University of
Pennsylvania Health System.
Penn's School of Medicine is ranked #2 in the nation for receipt
of NIH research funds; and ranked #3 in the nation in U.S. News
& World Report's most recent ranking of top research-oriented
medical schools. Supporting 1,400 fulltime faculty and 700 students,
the School of Medicine is recognized worldwide for its superior
education and training of the next generation of physician-scientists
and leaders of academic medicine.
The University of Pennsylvania Health System includes three hospitals,
all of which have received numerous national patient-care honors [Hospital
of the University of Pennsylvania; Pennsylvania Hospital, the nation's
first hospital; and Penn Presbyterian Medical Center]; a faculty practice
plan; a primary-care provider network; two multispecialty satellite
facilities; and home care and hospice.
Penn Medicine is one of the world’s leading academic medical centers, dedicated to the related missions of medical education, biomedical research, excellence in patient care, and community service. The organization consists of the University of Pennsylvania Health System and Penn’s Raymond and Ruth Perelman School of Medicine, founded in 1765 as the nation’s first medical school.
The Perelman School of Medicine is consistently among the nation's top recipients of funding from the National Institutes of Health, with $550 million awarded in the 2022 fiscal year. Home to a proud history of “firsts” in medicine, Penn Medicine teams have pioneered discoveries and innovations that have shaped modern medicine, including recent breakthroughs such as CAR T cell therapy for cancer and the mRNA technology used in COVID-19 vaccines.
The University of Pennsylvania Health System’s patient care facilities stretch from the Susquehanna River in Pennsylvania to the New Jersey shore. These include the Hospital of the University of Pennsylvania, Penn Presbyterian Medical Center, Chester County Hospital, Lancaster General Health, Penn Medicine Princeton Health, and Pennsylvania Hospital—the nation’s first hospital, founded in 1751. Additional facilities and enterprises include Good Shepherd Penn Partners, Penn Medicine at Home, Lancaster Behavioral Health Hospital, and Princeton House Behavioral Health, among others.
Penn Medicine is an $11.1 billion enterprise powered by more than 49,000 talented faculty and staff.