 “We showed for the first time that clotting is reversible,” says John Weisel, Ph.D., professor of Cell and Developmental Biology, in contrast to a long-standing assumption that it isn’t. Weisel and colleagues showed how these sometimes dangerous knots of protein and cells are actually a dynamic, mutable structure this month in Scientific Reports, a new Nature journal. Clotting is both a necessary function to stem blood loss, but in other dire circumstances can ultimately cause death.
“We showed for the first time that clotting is reversible,” says John Weisel, Ph.D., professor of Cell and Developmental Biology, in contrast to a long-standing assumption that it isn’t. Weisel and colleagues showed how these sometimes dangerous knots of protein and cells are actually a dynamic, mutable structure this month in Scientific Reports, a new Nature journal. Clotting is both a necessary function to stem blood loss, but in other dire circumstances can ultimately cause death.
In the last month since the paper has been online it has already risen to be the fifth-most-read paper in the journal in the past four weeks.
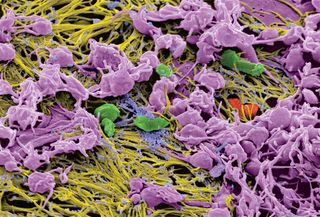
What makes this seemingly simple finding about the reversibility of clots so astounding is that researchers have tacitly assumed that clots and thrombi are stable structures until they fulfill their functions and are digested by the body. On the contrary, the Penn team found, using “fluorescence recovery after photobleaching,” that fibrin molecules come and go in clots, making them dynamic structures.
Clots are a network of fibers made up of fibrin molecules together with blood platelets, while thrombi are clots where they don’t belong that block the flow of blood. Photobleaching is a photochemical destruction of a dye used in microscopy as a way to study the motion of molecules that are tagged with fluorescent labels. In the conventional view of clotting, it would be expected that there would be no recovery of fluorescently labeled fibrin molecules after bleaching, but the observed recovery demonstrates that fibrin molecules are coming off adjacent parts of clot fibers and re-binding to the bleached area of the clot, Weisel explains.
“In other words, there are always some molecules coming off and going back on to the clot,” notes Weisel. This makes fibrin an equilibrium polymer, a term originally coined by Albert Einstein to describe other, non-biological polymers that are not permanent structures but display a balance between assembly and disassembly.
Indeed, fibrin is a remarkably versatile polymer. On one hand, fibrin is necessary to stem the loss of blood at an injury site. On the other, fibrin provides a scaffold for thrombi that block blood vessels and cause heart attacks and strokes.
“The body makes fibrin into a polymer in a specific and controlled way,” says Weisel. Think of it as a process that converts soluble fibrinogen molecules into insoluble fibrin molecules that interact in a knob-into-hole manner to form a network that is the basis of clots and thrombi.”
The team found that a peptide comprising the knobs of the fibrin polymerization process can compete for the holes and dissolve a preformed fibrin clot with striking rearrangements in the clot structure. Prashant Purohit, Ph.D., professor of Mechanical Engineering and Applied Mechanics at Penn, developed a mathematical model that made it possible to understand the turnover of fibrin molecules coming on and off a clot structure.
The results imply that clots or thrombi in the body are more dynamic structures than previously believed. What’s more, this process of turnover may account in part for embolization, which is the breaking away of portions of a clot that can travel through the blood vessels, causing damage if they lodge in small vessels of the brain or lungs.
“Since these results show that the equilibrium in clots can be shifted toward dissociating and disassembly of the fibrin molecules, they suggest a target for therapeutic intervention, seeking drugs that may push the reaction in this direction and potentially alleviating thrombosis without inducing bleeding,” says Weisel.
Chernysh IN, Nagaswami C, Purohit PK, & Weisel JW (2012). Fibrin clots are equilibrium polymers that can be remodeled without proteolytic digestion. Scientific reports, 2 PMID: 23170200 