| > |
Researchers at the Abramson
Family Cancer Research Institute of the University of Pennsylvania and
the Dana-Farber Cancer Institute describe a new candidate
breast-cancer susceptibility gene: the Rap80 gene
is required for the normal DNA-repair function of the well-known
breast cancer gene BRCA1. |
| > |
In this study, the researchers found
Rap80 binds to the region of the BRCA1 protein that is necessary
for recognizing sites of DNA damage. |
| > |
Therefore, Rap80, by interacting with
a BRCA1 region that is essential for BRCA tumor suppression,
now becomes a candidate to investigate as another breast
cancer disease gene in families that do not have BRCA1 and BRCA2 mutations,
but have a history of breast and/or ovarian cancer |
| > |
These findings are published in this
week’s
issue of Science |
(PHILADELPHIA) – Researchers at the Abramson
Family Cancer Research Institute of the University of Pennsylvania and the Dana-Farber
Cancer Institute describe in this week’s issue of Science a new candidate breast-cancer susceptibility gene. The Rap80 gene
is required for the normal DNA-repair function of the well-known
breast cancer gene BRCA1.
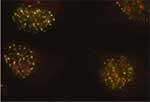 |
BRCA1 and Rap80 are recruited to the same structures at DNA damage sites in human cells treated with ionizing radiation.
Click on thumbnail
to view full-size images |
Cancer-causing mutations in the BRCA1 protein cause it to fail
to bind to the Rap80 protein. Consequently, BRCA1 is unable to
identify DNA damage sites in the genome. When BRCA1 fails to fix
DNA damage, cancer-causing mutations accumulate, spawning the development
of breast and ovarian malignancies.
“With this current discovery, we have made significant new
insights into the molecular mechanism by which BRCA1 recognizes
sites of DNA damage that breast-cancer-causing mutated forms of
BRCA1 cannot recognize,” says co-senior author Roger
Greenberg MD, PhD, Assistant Professor of Cancer Biology at Penn. “Now
we have gained a partial understanding of the molecular basis between
cancer-causing BRCA1 failures to fix DNA damage versus normal BRCA1’s
ability to fix DNA damage.”
In this study, the researchers found Rap80 binds to the region
of the BRCA1 protein that is necessary for recognizing sites of
DNA damage. In the 1990s, investigators discovered that BRCA1 was
involved in DNA repair by maintaining the normal number and structure
of chromosomes. DNA breaks that aren't repaired can lead to cancer
by increasing the rate of mutations, cancer-causing changes in
the gene sequence.
More specifically, modification of proteins in the cell nucleus – by
another protein called ubiquitin – that are tightly bound
to DNA are responsible for signaling BRCA1 via Rap80 to action.
Rap80 binds to specific types of ubiquitin that concentrate at
DNA damage sites, enabling BRCA1 to be recruited to sites of damage.
BRCA1 and BRCA2 mutations account for less than 50 percent of
inherited breast cancer. “The genetic basis of breast cancers
in other families has been largely unknown,” explains Greenberg. “These
families aren’t able to make informed choices about screening
and treatment, prophylactic or otherwise, the way the BRCA families
can.”
Researchers from multiple labs, including Penn and those led by
co-senior author David
M. Livingston at Dana-Farber, are finding
that many of these non-BRCA families have mutations in genes that
have a relationship with BRCA1. Many of the genes that encode these
proteins are also altered in familial breast cancer.
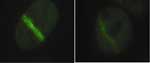 |
Depletion of Rap80 from human
cells (right panel) reduces the ability of BRCA1 to
migrate to laser-induced DNA damage sites as compared to
control cells (left panel).
Click on thumbnail
to view full-size images |
“Thus Rap80, by interacting with a BRCA1 region that is
essential for BRCA tumor suppression, now becomes a candidate to
investigate as another breast cancer disease gene in families that
do not have BRCA1 and BRCA2 mutations, but have a history of breast
and/or ovarian cancer,” says Greenberg. “In collaboration
with other researchers we are currently looking to see if families
that have a history of breast cancer, but lack BRCA1 and BRCA2 mutations, have any gene sequence changes in Rap80.”
This research was funded by the National
Cancer Institute and
startup funds from the Abramson Family Cancer Research Institute.
Co-authors are Bijan Sobhian, David M. Livingston, Aedín
C. Culhane, Lisa Moreau, and Bing, Xia from Dana Farber, and Genze
Shao and Dana R. Lilly from Penn.
###
PENN Medicine is a $2.9 billion enterprise
dedicated to the related missions of medical education, biomedical
research, and high-quality patient care. PENN Medicine consists
of the University of Pennsylvania School of Medicine (founded in
1765 as the nation's first medical school) and the University of
Pennsylvania Health System.
Penn's School of Medicine is ranked #2 in the nation for receipt
of NIH research funds; and ranked #3 in the nation in U.S. News
& World Report's most recent ranking of top research-oriented
medical schools. Supporting 1,400 fulltime faculty and 700 students,
the School of Medicine is recognized worldwide for its superior
education and training of the next generation of physician-scientists
and leaders of academic medicine.
The University of Pennsylvania Health System includes three hospitals,
all of which have received numerous national patient-care honors [Hospital
of the University of Pennsylvania; Pennsylvania Hospital, the nation's
first hospital; and Penn Presbyterian Medical Center]; a faculty practice
plan; a primary-care provider network; two multispecialty satellite
facilities; and home care and hospice.
Penn Medicine is one of the world’s leading academic medical centers, dedicated to the related missions of medical education, biomedical research, excellence in patient care, and community service. The organization consists of the University of Pennsylvania Health System and Penn’s Raymond and Ruth Perelman School of Medicine, founded in 1765 as the nation’s first medical school.
The Perelman School of Medicine is consistently among the nation's top recipients of funding from the National Institutes of Health, with $550 million awarded in the 2022 fiscal year. Home to a proud history of “firsts” in medicine, Penn Medicine teams have pioneered discoveries and innovations that have shaped modern medicine, including recent breakthroughs such as CAR T cell therapy for cancer and the mRNA technology used in COVID-19 vaccines.
The University of Pennsylvania Health System’s patient care facilities stretch from the Susquehanna River in Pennsylvania to the New Jersey shore. These include the Hospital of the University of Pennsylvania, Penn Presbyterian Medical Center, Chester County Hospital, Lancaster General Health, Penn Medicine Princeton Health, and Pennsylvania Hospital—the nation’s first hospital, founded in 1751. Additional facilities and enterprises include Good Shepherd Penn Partners, Penn Medicine at Home, Lancaster Behavioral Health Hospital, and Princeton House Behavioral Health, among others.
Penn Medicine is an $11.1 billion enterprise powered by more than 49,000 talented faculty and staff.