| > |
Immune cells first need to
recognize infecting microbes, then divide and differentiate into
the type of immune cells that fight off infection. |
| > |
Researchers at the University
of Pennsylvania School of Medicine have
discovered that proteins that regulate cell fate – whether
to become the kamikaze T-cell or to stay like the mother cell
and retain memory of the microbe invader – ended up in
one or the other of the two of the daughter cells, but not
both. Many immunologists thought that when immune cells receive
a signal, they all become one type of daughter cell. |
| > |
The researchers studied the immune response
to the bacteria Listeria in
a mouse model. By extracting Listeria-specific
T-cells from the mouses’ spleen and
using a special dye label, the investigators were able to
track the T-cells’ first divisions upon infection,
telling the difference between the original mother T-cell
and subsequent daughter cells. |
| > |
The research appears this week in an
early online issue of Science. |
(PHILADELPHIA) – How do immune
cells decide
to respond to invading microbes by either fighting to the death
or becoming the body’s memory
for future infections? Researchers at the University
of Pennsylvania School of Medicine have discovered that immune cells can differ
in their inheritance of molecules that regulate cell fate, and
therefore what role they play in fighting infection. The research
appears this week in an early online issue of Science.
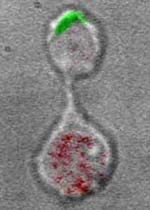 |
Listeria-specific T-cell in final stages
of cellular division
Click on thumbnail
to view full-size image |
“These findings will change the way we look at how vaccines work,” says lead author Steven Reiner,
MD, Professor of Medicine. “Until
now, we have pretty much been in the dark as to how we generate
the spectrum of immune cells that are needed to fight off infections.
We've always known the end result – that more than one type
of cell is needed – but not the initial events that generate
cell diversity. Now we have information that may one day be applied
to a more targeted approach to developing vaccines.”
Immune cells first need to recognize infecting microbes, then
divide and differentiate into the type of immune cells that fight
off infection. But how, after replicating, do the new daughter
cells know to become fighter cells that are expendable – in
effect kamikazes – and yet reserve some cells to “remember” the
invading microbe for future infections?
“That's the conundrum of the immune system and the extraordinary
potential of a stem
cell,” explains Reiner. After the initial
infection, some of the descendents of an immune cell reacting to
a microbe must become the soldiers that fight the infection and
die; but some, instead, regenerate to take the place of the mother
cell, leaving an immunologic memory to recognize the invading microbe
in the future.
Given the simplicity of this scenario, surprisingly few examples
have been well-characterized that show how daughter cells can be
different in their inheritance of molecules that regulate cell
fate. The Science paper establishes how immune cells can achieve
this asymmetric or unequal inheritance.
The researchers studied the immune response to the bacteria Listeria in a mouse model. They transplanted T-cells specific to Listeria into the mice and then infected the mouse with Listeria. By extracting Listeria-specific T-cells from the mouses’ spleen and using
a special dye label, the investigators were able to track the T-cells’ first
divisions upon infection, telling the difference between the original
mother T-cell and subsequent daughter cells.
In T-cells on the verge of division, and which were the first
responding to Listeria, the researchers tracked different
types of molecules that each daughter cell retained after division.
Proteins that regulate cell fate – whether to become the
kamikaze T-cell or to stay like the mother cell and retain memory
of the microbe invader – ended up in one or the other of
the two of the daughter cells, but not both. “We saw a lopsided
immune response within the first cell division,” says Reiner.
Reiner and colleagues say that this line of research will change
how scientists think about something as basic as how our bodies
react to infection. "This will shock many immunologists because
many of them think that when immune cells receive a signal, they
all change one way or another, not in a way that makes two different
daughter cells,” says Reiner. “In terms of immunity
and vaccines, we now understand how immune cells can generate this
asymmetric cell fate.”
This study was funded by the National
Institutes of Health. Co-authors
in addition to Reiner are John T. Chang, Vikram R. Palanivel, Ichiko
Kinjyo, Felix Schambach, Andrew M. Intlekofer, Arnob Banerjee,
Sarah A. Longworth, Kristine E. Vinup, and Jordan S. Orange, all
from Penn. Paul
Mrass and Wolfgang
Weninger (The
Wistar Institute,
Philadelphia); Jane
Oliaro and Sarah M. Russell (Peter
MacCallum Cancer Centre, Victoria Australia); and Nigel
Killeen (UC-San
Francisco)
are also co-authors.
###
PENN Medicine is a $2.9 billion enterprise
dedicated to the related missions of medical education, biomedical
research, and high-quality patient care. PENN Medicine consists
of the University of Pennsylvania School of Medicine (founded in
1765 as the nation's first medical school) and the University of
Pennsylvania Health System.
Penn's School of Medicine is ranked #2 in the nation for receipt
of NIH research funds; and ranked #3 in the nation in U.S. News
& World Report's most recent ranking of top research-oriented
medical schools. Supporting 1,400 fulltime faculty and 700 students,
the School of Medicine is recognized worldwide for its superior
education and training of the next generation of physician-scientists
and leaders of academic medicine.
The University of Pennsylvania Health System includes three hospitals,
all of which have received numerous national patient-care honors [Hospital
of the University of Pennsylvania; Pennsylvania Hospital, the nation's
first hospital; and Penn Presbyterian Medical Center]; a faculty practice
plan; a primary-care provider network; two multispecialty satellite
facilities; and home care and hospice.
Penn Medicine is one of the world’s leading academic medical centers, dedicated to the related missions of medical education, biomedical research, excellence in patient care, and community service. The organization consists of the University of Pennsylvania Health System and Penn’s Raymond and Ruth Perelman School of Medicine, founded in 1765 as the nation’s first medical school.
The Perelman School of Medicine is consistently among the nation's top recipients of funding from the National Institutes of Health, with $550 million awarded in the 2022 fiscal year. Home to a proud history of “firsts” in medicine, Penn Medicine teams have pioneered discoveries and innovations that have shaped modern medicine, including recent breakthroughs such as CAR T cell therapy for cancer and the mRNA technology used in COVID-19 vaccines.
The University of Pennsylvania Health System’s patient care facilities stretch from the Susquehanna River in Pennsylvania to the New Jersey shore. These include the Hospital of the University of Pennsylvania, Penn Presbyterian Medical Center, Chester County Hospital, Lancaster General Health, Penn Medicine Princeton Health, and Pennsylvania Hospital—the nation’s first hospital, founded in 1751. Additional facilities and enterprises include Good Shepherd Penn Partners, Penn Medicine at Home, Lancaster Behavioral Health Hospital, and Princeton House Behavioral Health, among others.
Penn Medicine is an $11.1 billion enterprise powered by more than 49,000 talented faculty and staff.