| > |
Researchers at the University
of Pennsylvania School of Medicine have discovered
a molecular link between the cell’s two major pathways
for breaking down proteins and have succeeded in using
this link to rescue neurodegenerative diseases in a simple
animal model. |
| > |
The cell has two internal pathways for
breaking down proteins. The ubiquitin-proteasome pathway
marks unwanted proteins with ubiquitin tags and shuttles
them for rapid breakdown to a complicated structure called
the proteasome. The second is the autophagy-lysosomal system,
a more general process in which proteins are surrounded by
membranes inside the cell for bulk digestion. |
| > |
The researchers discovered that making
the lysosomal system more or less active dramatically influenced
the severity of neurodegeneration. |
| > |
The study appears this week in Nature. |
(PHILADELPHIA) – Many age-related neurological diseases
are associated with defective proteins accumulating in nerve
cells,
suggesting that the cell’s
normal disposal mechanisms are not operating correctly. Now, researchers
at the University of Pennsylvania School of
Medicine have discovered
a molecular link between the cell’s two major pathways for
breaking down proteins and have succeeded in using this link to
rescue neurodegenerative diseases in a simple animal model. The
study appears this week in Nature.
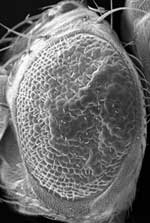 |
Eye in fruit fly model of Alzheimer's disease
appears rough and deformed.
Click on thumbnail
to view full-size images |
The cell has two internal pathways for breaking down proteins.
The ubiquitin-proteasome pathway marks unwanted proteins with ubiquitin tags and shuttles
them for rapid breakdown to a complicated structure called the
proteasome. The second is the autophagy-lysosomal system,
a more general process in which proteins are surrounded by membranes inside the cell for bulk digestion.
“The dogma has been that the autophagy-lysosomal and the
proteasomal systems are trains that run on different tracks, with
similar purposes, but no point of intersection,” explains
senior author J. Paul Taylor, MD, PhD, Assistant Professor of Neurology. “The
new finding directly challenges this thinking by showing that one
system can be induced to compensate for the other. Cells are able
to shift proteins between the systems. We think that this molecular
link can be used to benefit a wide variety of neurodegenerative
diseases because accumulation of toxic proteins is a common underlying
feature of age-related neurodegeneration.”
Taylor and his group study fruit flies in which the proteasome
is disabled by a genetic mutation, which results in neurodegeneration.
They use the fly eye, a neuron-rich tissue, as a surrogate for
the brain because it is easy to visualize. They discovered that
making the lysosomal system more or less active dramatically influenced
the severity of neurodegeneration.
“We found that whenever we knocked the lysosome system down,
neurodegeneration always got worse,” says Taylor. “Then
when we activated the autophagy system by feeding the flies a drug
called rapamycin, neurodegeneration was prevented.” The accumulated
misfolded proteins were cleared out by the lysosome system. “Then
we knew that this system can compensate for the impaired proteasome
function, which in itself tells us that the two pathways intersect,” says
Taylor. “The question was, ‘How is this working?’”
The Role of HDAC6
“That’s where the power of fruit flies comes in,” Taylor
explains. “We can use fruit flies to rapidly screen through
many genes to find the one we’re interested in. In the process
of screening, our attention was drawn to HDAC6 because we already
knew that it could bind to ubiquitin-tagged proteins and transport
them within the cell. So we wondered, could HDAC6 be the link?”
Taylor’s group showed that if the HDAC6 gene is knocked
out, inducing autophagy no longer rescues the fly eyes from neurodegeneration.
Therefore, autophagy requires HDAC6 to work. They also showed that
by simply expressing extra HDAC6, neurodegeneration was prevented
in flies with proteasome impairment. Taylor’s group then
moved on to fly models of human neurodegenerative disease and showed
that they, too, are rescued by over-expression of HDAC6.
Therefore, the researchers suggest that the level of the HDAC6
in a cell regulates its sensitivity to accumulation of misfolded
proteins, and that increasing the activity of HDAC6 can prevent
the degeneration normally associated with accumulating old, damaged
proteins. The researchers suggest further that when proteasomes
are impaired or overwhelmed, which leads to accumulation of defective
proteins, HDAC6 facilitates delivery to the autophagy-lysosomal
system for degradation. “That’s how we think HDAC6
links the two systems,” says Taylor.
Taylor and his team are now testing the ability of HDAC6 to
prevent neurodegeneration in several mouse models, including motor
neuron disease, Parkinson’s disease, and Huntington’s
disease. They are also attempting to identify pharmacologic approaches
to augmenting HDAC6 activity.
Penn co-authors are Udai
Bhan Pandey, Zhiping
Nie, Brett
A. McCray,
Gillian P.
Ritson, Natalia
B. Nedelsky, and Stephanie Schwartz.
This work was funded by the Kennedy’s Disease Association,
the Morton Reich Research Fund, and the National
Institute of Neurological Disorders and Stroke.
###
PENN Medicine is a $2.9 billion enterprise
dedicated to the related missions of medical education, biomedical
research, and high-quality patient care. PENN Medicine consists
of the University of Pennsylvania School of Medicine (founded in
1765 as the nation's first medical school) and the University of
Pennsylvania Health System.
Penn's School of Medicine is ranked #2 in the nation for receipt
of NIH research funds; and ranked #3 in the nation in U.S. News
& World Report's most recent ranking of top research-oriented
medical schools. Supporting 1,400 fulltime faculty and 700 students,
the School of Medicine is recognized worldwide for its superior
education and training of the next generation of physician-scientists
and leaders of academic medicine.
The University of Pennsylvania Health System includes three hospitals,
all of which have received numerous national patient-care honors [Hospital
of the University of Pennsylvania; Pennsylvania Hospital, the nation's
first hospital; and Penn Presbyterian Medical Center]; a faculty practice
plan; a primary-care provider network; two multispecialty satellite
facilities; and home care and hospice.
Penn Medicine is one of the world’s leading academic medical centers, dedicated to the related missions of medical education, biomedical research, excellence in patient care, and community service. The organization consists of the University of Pennsylvania Health System and Penn’s Raymond and Ruth Perelman School of Medicine, founded in 1765 as the nation’s first medical school.
The Perelman School of Medicine is consistently among the nation's top recipients of funding from the National Institutes of Health, with $550 million awarded in the 2022 fiscal year. Home to a proud history of “firsts” in medicine, Penn Medicine teams have pioneered discoveries and innovations that have shaped modern medicine, including recent breakthroughs such as CAR T cell therapy for cancer and the mRNA technology used in COVID-19 vaccines.
The University of Pennsylvania Health System’s patient care facilities stretch from the Susquehanna River in Pennsylvania to the New Jersey shore. These include the Hospital of the University of Pennsylvania, Penn Presbyterian Medical Center, Chester County Hospital, Lancaster General Health, Penn Medicine Princeton Health, and Pennsylvania Hospital—the nation’s first hospital, founded in 1751. Additional facilities and enterprises include Good Shepherd Penn Partners, Penn Medicine at Home, Lancaster Behavioral Health Hospital, and Princeton House Behavioral Health, among others.
Penn Medicine is an $11.1 billion enterprise powered by more than 49,000 talented faculty and staff.