| > |
Aggressive
brain tumors (glioblastoma multiforme, or GBM) that do
not respond to traditional chemoradiation treatment may
respond to Cotara®. |
| > |
Cotara
is a monoclonal antibody that targets the DNA of cancer
cells and carries a radioactive isotope. |
| > |
Cotara
is delivered through a method called convection enhanced
delivery (CED), which uses a catheter to bypass the blood-brain
barrier and target the specific tumor site in the brain. |
| > |
The Hospital
of the University of Pennsylvania (HUP) is one
of four sites participating in the clinical trail of
Cotara. |
(PHILADELPHIA) – Physicians initially diagnosed
Phil Marfuta, 28, with tension
headaches, which seemed reasonable
to him since he is a busy graduate student studying physics at
Princeton University. However, as the days went on his headaches
did not subside, and when a CT
scan and an MRI revealed two tumors,
Phil underwent emergency surgery at the Hospital of the
University of Pennsylvania.
One of Phil’s tumors was a grade IV glioblastoma
multiforme (GBM), which is the most aggressive form of primary brain tumor.
Typically once diagnosed, the median survival time for a patient
with a GBM is 12 months.
“That kind of news is the kind you don’t want to have
to call your family and tell them about,” says Phil.
Phil’s tumors had stopped responding to traditional chemoradiation treatment and to a clinical trial that added an immune
modulator to his therapy. That’s when he learned of the Cotara® trial.
About Cotara® and the Clinical Trial
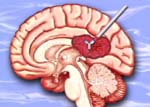 |
Cotara is delivered through a method called convection
enhanced delivery (CED).
Click on thumbnail
to view full-size image
|
Cotara is a monoclonal antibody that targets the DNA of cancer cells and carries a radioactive
isotope to them, literally destroying
the cancer cells from the inside out. GBMs are complicated
to treat because filaments of malignant
cells spread out like fingers
from the tumor and take root deep in the surrounding tissue, making
it difficult to remove them without damaging healthy brain cells. This
approach targets the cancer cells, while sparing healthy tissue
in the brain.
In November, 2006, Phil was treated in this clinical trial of
Cotara. “It involved
just catheters,
so compared to a craniotomy,
that sounded quite nice,” recalls Phil. To-date, Phil’s
GBM has been stabilized
HUP is one of four sites participating in the clinical trail of
Cotara, a biological “guided missile” for the treatment
of GBM, a deadly form of brain cancer that is newly diagnosed in
more than 10,000 Americans each year.
Phil feels very strongly about the importance of participating
in clinical trials and has even posted an account of his experience
on the Young Adults Surviving Glioblastoma’s Web site.
“I’m very passionate about science and I really enjoy
finding things out and experimenting. By participating in
this trial, somehow I feel like I’ve actually done something
tangible to help other people who are basically going through the
same thing that I am going through. That’s a small
thing, but to me, it’s a very active step and it makes me
feel good that there’s a contribution that I’ve made,” says
Phil. “I sincerely wish that people would take that
step and participate in these trials.”
A Different Approach for Treating Brain Tumors
“Based on the scientific rationale for Cotara, coupled with
some positive but preliminary indications from previous trials,
we believe it is important to study this approach as a new option
for this deadly disease,” says Kevin D. Judy, MD,
Associate Professor, Department
of Neurosurgery at Penn.
“One challenge in treating brain tumors is the difficulty
of delivering therapeutic agents to the brain through the blood-brain
barrier (BBB) – the physical shield that serves to protect
the brain and central
nervous system from potentially dangerous
or infectious agents,” says Myrna R. Rosenfeld, MD,
PhD, Chief of Penn’s Division
of Neuro-Oncology. Cotara
is delivered through a method called convection
enhanced delivery (CED), which uses a catheter to bypass the BBB and target the specific
tumor site in the brain. “This method enables us to
treat GBMs with local therapy that does not have side effects elsewhere
in the body,” says Robert Lustig, MD, Clinical
Associate Professor of Radiation
Oncology at Penn.
The Objective of the Clinical Trial
The Phase
2 clinical trial is designed to evaluate a single Cotara
infusion in patients with a first or second recurrence of GBM. The
primary endpoints of the trial are to confirm safety and determine
median survival time and median time to progression in patients
treated with Cotara. Cotara has been granted orphan
drug status and fast
track designation by the U.S. Food
and Drug Administration for the treatment of GBM.
Peregrine Pharmaceuticals,
Inc., developers of Cotara, is working
with the New Approaches to Brain
Tumor Therapy (NABTT) to conduct
the trial at Penn, Wake Forest University, Emory
University, and
the University of Alabama at Birmingham.
Editor's Notes: Dr. Judy, Dr. Rosenfeld, and
Dr. Lustig have no financial interest in Peregrine Pharmaceuticals,
Inc.
For additional information about the trial at Penn, call (215)
615-4590.
For information regarding the other sites, call (800) 694-5334
or visit www.clinicaltrials.gov/ct/show/NCT00128635?order=1
###
PENN Medicine is a $2.9 billion enterprise
dedicated to the related missions of medical education, biomedical
research, and high-quality patient care. PENN Medicine consists
of the University of Pennsylvania School of Medicine (founded in
1765 as the nation's first medical school) and the University of
Pennsylvania Health System.
Penn's School of Medicine is ranked #2 in the nation for receipt
of NIH research funds; and ranked #3 in the nation in U.S. News
& World Report's most recent ranking of top research-oriented
medical schools. Supporting 1,400 fulltime faculty and 700 students,
the School of Medicine is recognized worldwide for its superior
education and training of the next generation of physician-scientists
and leaders of academic medicine.
The University of Pennsylvania Health System includes three hospitals,
all of which have received numerous national patient-care honors [Hospital
of the University of Pennsylvania; Pennsylvania Hospital, the nation's
first hospital; and Penn Presbyterian Medical Center]; a faculty practice
plan; a primary-care provider network; two multispecialty satellite
facilities; and home care and hospice.
Penn Medicine is one of the world’s leading academic medical centers, dedicated to the related missions of medical education, biomedical research, excellence in patient care, and community service. The organization consists of the University of Pennsylvania Health System and Penn’s Raymond and Ruth Perelman School of Medicine, founded in 1765 as the nation’s first medical school.
The Perelman School of Medicine is consistently among the nation's top recipients of funding from the National Institutes of Health, with $550 million awarded in the 2022 fiscal year. Home to a proud history of “firsts” in medicine, Penn Medicine teams have pioneered discoveries and innovations that have shaped modern medicine, including recent breakthroughs such as CAR T cell therapy for cancer and the mRNA technology used in COVID-19 vaccines.
The University of Pennsylvania Health System’s patient care facilities stretch from the Susquehanna River in Pennsylvania to the New Jersey shore. These include the Hospital of the University of Pennsylvania, Penn Presbyterian Medical Center, Chester County Hospital, Lancaster General Health, Penn Medicine Princeton Health, and Pennsylvania Hospital—the nation’s first hospital, founded in 1751. Additional facilities and enterprises include Good Shepherd Penn Partners, Penn Medicine at Home, Lancaster Behavioral Health Hospital, and Princeton House Behavioral Health, among others.
Penn Medicine is an $11.1 billion enterprise powered by more than 49,000 talented faculty and staff.