| > |
Past studies in tissue culture
have suggested that one type of pancreas cell could be coaxed
to transform into insulin-producing islet cells. |
| > |
Researchers at the University
of Pennsylvania School of Medicine have demonstrated
that these pancreatic acinar cells do not become insulin-producing
cells in an animal model. |
| > |
The researchers did show that injured pancreatic cells
readily regenerate back into healthy acinar cells, which
has implications for treating cancer and inflammation of
the pancreas. |
| > |
The research also holds promise for new
techniques for pancreas cell manipulation. |
| > |
This study appears in the April issue
of The Journal of Clinical Investigation. |
(PHILADELPHIA) – Replacing faulty or missing cells with
new insulin-making cells has been the object of diabetes research
for the last decade. Past studies in tissue culture have suggested
that one type of pancreas cell could be coaxed to transform into
insulin-producing islet
cells.
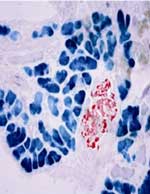 |
Mice cells with permanently and selectively
labeled pancreatic acinar cells (blue) and insulin-producing
cells (red)
Click on thumbnail
to view full-size image |
Now, researchers at the University
of Pennsylvania School of Medicine have demonstrated
that these pancreatic acinar cells do not become insulin-producing
cells in an animal model. However, they did show that injured
pancreatic cells readily regenerate back into healthy acinar
cells, which has implications for treating cancer and inflammation
of the pancreas. This study appears in the April issue of The Journal
of Clinical Investigation. The research also holds promise
for new techniques for pancreas cell manipulation.
The pancreas is made up of two compartments with different functions:
the islet compartment of insulin-producing beta
cells and the much
larger exocrine compartment composed of duct cells and acinar cells
that make and deliver enzymes to the intestine for digestion. Diabetes
is caused by the failure of the beta cells to make insulin, whereas
pancreatic cancer usually originates from the exocrine compartment.
Under certain conditions in tissue culture, acinar cells can synthesize insulin as well as amylase, a digestion enzyme.
“These findings have the potential to change the emphasis
in diabetic research as far as regenerating the pancreas is concerned,” says
lead author Doris Stoffers, MD, PhD, Assistant Professor of Medicine.
Evidence from Stoffers’ group and other groups is pointing
to the beta cell itself as the most promising source for generating
new beta cells. The focus of research is now shifting toward the
direct stimulation of islet cell growth in live animals. In contrast,
once acinar cells are removed from the organism and placed into
culture, they may have greater potential to change into other cell
types, including beta cells. As a result, Stoffers’ animal
model and technical approach is currently being used by other groups
in the United States, Europe, and China to determine conditions
under which acinar cells can take on the features of duct cells
and beta cells.
The Penn team engineered mice with a special marker that permanently
and selectively labels only pancreatic acinar cells. The mice were
then subjected to pancreatic injury by chemicals or surgery. The
pancreas was allowed to heal or regenerate itself, and the specific
acinar cell marker was followed microscopically in thin slices
of pancreatic tissue. “It is very clear that the acinar and
islet compartments remain separate during regeneration in a live
animal,” says Stoffers.
“Although our work shows that acinar cells do not contribute
to the insulin-producing compartment of the pancreas in an animal
model, it is possible that other strategies might be successful
in generating the islet cells,” says Stoffers. Ongoing research
is examining whether acinar cells from the mice used in this study
can be induced to make insulin in tissue culture. “The hope
is that these acinar cells would continue to make insulin after
being transplanted back into the mouse,” says Stoffers.
The American Diabetes Association and the National
Institute of Diabetes and Digestive and Kidney Diseases funded the research.
Coauthors of the study are Biva
M. Desai, Jennifer
Oliver-Krasinski,
Diva
D. De Leon, Cyrus
Farzad, and Nankang Hong, all from Penn
along with Steven
D. Leach, from the Johns
Hopkins University School of Medicine.
###
PENN Medicine is a $2.9 billion enterprise
dedicated to the related missions of medical education, biomedical
research, and high-quality patient care. PENN Medicine consists
of the University of Pennsylvania School of Medicine (founded in
1765 as the nation's first medical school) and the University of
Pennsylvania Health System.
Penn's School of Medicine is ranked #2 in the nation for receipt
of NIH research funds; and ranked #3 in the nation in U.S. News
& World Report's most recent ranking of top research-oriented
medical schools. Supporting 1,400 fulltime faculty and 700 students,
the School of Medicine is recognized worldwide for its superior
education and training of the next generation of physician-scientists
and leaders of academic medicine.
The University of Pennsylvania Health System includes three hospitals,
all of which have received numerous national patient-care honors [Hospital
of the University of Pennsylvania; Pennsylvania Hospital, the nation's
first hospital; and Penn Presbyterian Medical Center]; a faculty practice
plan; a primary-care provider network; two multispecialty satellite
facilities; and home care and hospice.
Penn Medicine is one of the world’s leading academic medical centers, dedicated to the related missions of medical education, biomedical research, excellence in patient care, and community service. The organization consists of the University of Pennsylvania Health System and Penn’s Raymond and Ruth Perelman School of Medicine, founded in 1765 as the nation’s first medical school.
The Perelman School of Medicine is consistently among the nation's top recipients of funding from the National Institutes of Health, with $550 million awarded in the 2022 fiscal year. Home to a proud history of “firsts” in medicine, Penn Medicine teams have pioneered discoveries and innovations that have shaped modern medicine, including recent breakthroughs such as CAR T cell therapy for cancer and the mRNA technology used in COVID-19 vaccines.
The University of Pennsylvania Health System’s patient care facilities stretch from the Susquehanna River in Pennsylvania to the New Jersey shore. These include the Hospital of the University of Pennsylvania, Penn Presbyterian Medical Center, Chester County Hospital, Lancaster General Health, Penn Medicine Princeton Health, and Pennsylvania Hospital—the nation’s first hospital, founded in 1751. Additional facilities and enterprises include Good Shepherd Penn Partners, Penn Medicine at Home, Lancaster Behavioral Health Hospital, and Princeton House Behavioral Health, among others.
Penn Medicine is an $11.1 billion enterprise powered by more than 49,000 talented faculty and staff.