 It's not every day you see a worm on the cover of Science magazine. But a recent issue featured a member of the helminth parasite family to highlight back-to-back articles investigating how worms can influence immune responses to viral infection. David Artis, PhD, professor of Microbiology and his team, including first author Lisa Osborne, PhD, demonstrated that mice already infected with parasitic helminths were worse at fighting viral infection. They met with me to explain this line of work and what it may mean for inflammatory diseases and better responses to vaccines.
It's not every day you see a worm on the cover of Science magazine. But a recent issue featured a member of the helminth parasite family to highlight back-to-back articles investigating how worms can influence immune responses to viral infection. David Artis, PhD, professor of Microbiology and his team, including first author Lisa Osborne, PhD, demonstrated that mice already infected with parasitic helminths were worse at fighting viral infection. They met with me to explain this line of work and what it may mean for inflammatory diseases and better responses to vaccines.
(That photo above is a color-enhanced scanning electron micrograph of the helminth parasite Trichuris muris in the mouse intestine, shown in a Warholesque montage.)
Helminth infections are included in the category of “neglected tropical diseases,” typically underfunded and understudied relative to the burden of those affected. These “neglected” parasites infect approximately 2 billion people worldwide, roughly one-third of the world’s population. Although these agents don’t kill as many people as TB or malaria, epidemiologic and clinical data suggests helminth infection can impact progression of other infections, such as HIV, and induce a weak response to vaccination, especially in children. Therefore, understanding the impact of these parasites on the host immune response could have far-reaching implications.
In the Science papers, investigators at Penn and Washington University (WU) addressed the issue of what happens when parasitic worms and viruses simultaneously infect mice. Artis and Osborne demonstrated that previously existing helminth infection impairs virus clearance, and the WU group found that parasite co-infection woke up a dormant herpes virus in mice. The work from both groups is highlighted in a Perspective in Science and an editor’s summary, which conclude that “worms skewed the immune response so that the immune cells and the molecules they secreted created an environment favorable for the worm at the expense of antiviral immunity.”
 Helminths induce potent effects on the immune system, but whether these effects are mediated directly by the host or indirectly through changes in the microbiome – namely harmless commensal bacteria -- is not known. Since helminth worms live in the gut right next to beneficial commensal bacteria, the Penn team investigated whether helminth infection dampened host immune responses through “re-setting” or restructuring the community of commensal bacteria. Although this idea has been previously suggested, little data has so far examined this question directly.
Helminths induce potent effects on the immune system, but whether these effects are mediated directly by the host or indirectly through changes in the microbiome – namely harmless commensal bacteria -- is not known. Since helminth worms live in the gut right next to beneficial commensal bacteria, the Penn team investigated whether helminth infection dampened host immune responses through “re-setting” or restructuring the community of commensal bacteria. Although this idea has been previously suggested, little data has so far examined this question directly.
To do this, researchers need germ-free mice that have been born and raised in the complete absence of live commensal bacteria. Artis and Osborne did this, and showed that helminth-infected mice had poor immune responses following viral infection, regardless of whether commensal bacteria were present. These findings -- showing that helminth infection diminished antiviral immunity in microbe-free and non-microbe-free mice – suggests that helminth infection can influence immune cells directly. In fact, they showed that helminth-induced alternative activation of macrophages was associated with the impaired immune response to viruses. In particular, neutralization of Ym1, a molecule associated with alternatively activated macrophages, could partially restore antiviral immunity. They concluded that helminth-induced immune suppression occurs independently of changes in gut bacteria but is dependent on Ym1.
This sentence at the end of the Osborne/Artis paper prompted me to ask them about clinical applications of studying worm infections:
The immunomodulatory role for the Ym1 pathway could have implications for the development of helminth-base therapies for multiple chronic inflammatory diseases as well as improved vaccination strategies in helminth-infected individuals.
They explained two implications. Kids in less industrialized countries have impaired responses to vaccination. For example, the rotavirus vaccine is greater than 90 percent effective in industrialized countries and only about 50 to 60 percent effective in less industrialized countries. Artis and Osborne surmise that physicians one day might be able to increase the responsiveness to vaccines by blocking Ym1 or related molecules. A lot more work needs to be done, but at least for now, this field has a new hypothesis to test that may help explain these decreased responses to vaccines.
The second implication is related to helminths’ ability to shut down specific aspects of the immune system to survive, without generally killing the host. Artis describes that as a possible example of co-evolved mutualism. Using this family of worms has been proposed to dampen the activation of immune cells in the context of inflammatory diseases in which the immune system goes into overdrive and has been proposed or tested in various settings including IBD, arthritis, asthma and MS.
In fact, clinical trials investigating the safety and efficacy of helminth immunotherapy for IBD, MS and asthma using live, controlled helminth infections are ongoing. The increased prevalence of these inflammatory diseases in industrialized countries where citizens do not have helminth infections lends support to testing worm-based immunotherapeutics, say some researchers.
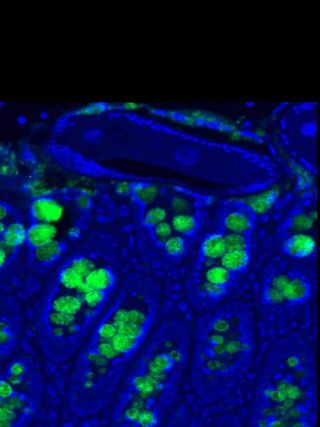
Caption for second image: Histologic section of murine intestine infected with the helminth parasite Trichuris muris. Blue marks cell nuclei and green marks helminth-induced Realm expression (image provided by Meera Nair and David Artis).